Plasmid vectors: the mystery vector
Plasmid vectors are key to modern biological research but can be prone to misidentification and loss through a variety of laboratory mishaps. This post outlines some easy steps you can take to confidently recover lost or misidentified plasmid vectors.

Plasmid vectors are an essential, ubiquitous, and powerful tool in modern life science research, yet they can often be a point of difficulty or failure in key experiments. While the classic plasmid preparation is a valuable and illustrative mainstay of the undergraduate biochemistry laboratory, the modern academic and industrial research environment dictates that researchers maximize the use of precious time and funding by ensuring the efficient creation and use of these basic tools. Many experimental applications require the reliable and regular use of relatively large quantities of high-quality vector DNA, including sterile and endotoxin-free preparations in cell culture.
Furthermore, the use of low-quality DNA, the incorrect vector, or a vector with an incorrect sequence in an experiment can prove disastrous and cause significant delay and frustration. Thankfully, the application of the tips and tricks presented in this blog post series, either in your own lab or on your behalf by Proxima, can ensure inexpensive, reliable, repeatable, and hassle-free creation, maintenance, isolation, and application of plasmid vectors in your research. Today we’ll begin where many of us have had to begin our plasmid work in the lab – the mystery vector.
Identifying the mystery vector
We’ve all been there. Perhaps a former lab member had less than ideal record-keeping, perhaps the label on a tube has become faded or blurred, or perhaps some documentation or a plasmid map has been misplaced. Worse yet; a plasmid stock has inadvertently been used up leaving only an empty tube; or a freezer failure has resulted in racks and racks of thawed, and quite ruined, glycerol stocks. Whatever the cause, the outcome is the same, a vector critical for our research is lost or its sequence accuracy and identity are dubious. Fear not! There are easy ways to identify key features of your vector and recover it – so don’t throw out that empty tube and save those ruined glycerol stocks!
Recovering vector DNA
Let’s first consider the case of the empty tube or dead glycerol stock. Simply rinsing the inside of an apparently empty tube with ten to twenty microlitres of nuclease-free water followed by gentle heating (55 °C, 30 min, with intermittent vortexing) will typically elute and recover sufficient residual DNA from the walls of the tube for analyses and transformation as described below. Glycerol stocks may fail for several reasons, and cells may be dead even though the stock has remained frozen. More commonly, freezer failure or accidental or frequent thawing results in a stock from which live cells cannot be recovered. Although in both cases the cells are dead the vector DNA typically remains substantially intact. Simply spinning down the solution and running the resulting cell pellet through a plasmid miniprep kit will generally yield more than enough DNA for transformation and analysis.
Stocks which have been thawed or damaged to the point where the cells are no longer intact can still be used to recover the vector. In this case, a genomic or total DNA extraction kit is used to ensure the recovery of all DNA from the mixture. Following analysis, the vector is recovered by transformation and selection with the appropriate antibiotic. Using this approach, we have successfully recovered a plasmid vector from a glycerol stock which was dropped on the floor and lost under a warm fridge for over a year!
Determining selectable markers
The next hurdle in recovering a vector where very little is available (< 10 ng) is knowing what selectable marker is present so the plasmid can be transformed, and sufficient material prepared for further analyses. If the selectable marker is unknown, we apply a qPCR-based approach using primers directed at common selectable marker genes such as AmpR and KanR. This method is effective for even sub-nanogram quantities of vector, and if the sample is particularly precious the PCR reaction itself may be used for transformation. Regular PCR followed by gel electrophoresis also works but involves an extra step. If sample quantity is especially limited, loop-mediated isothermal amplification (LAMP) can identify the selectable marker present using DNA inputs as low as one femtogram.
Having identified the selectable marker in your vector the next challenge is transformation. Many companies offer ultra-high competency cells, and this is the time to purchase some and use them, even if you are accustomed to making your own. Follow the protocol to maximize transformation and do a few trial transformations with 1 ng of a known good vector to develop competency with transformation using small amounts of DNA. A big factor in success is repetition and familiarity with handling very small quantities of DNA, and with that experience, we have been able to reliably transform picogram quantities of vectors.
Elucidating sequence and structure
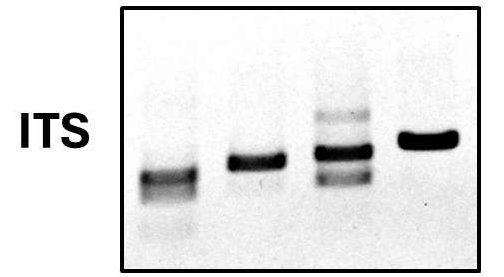
Having grown up or otherwise obtained a significant quantity of your mystery vector the final step is elucidating its sequence and structure. The most direct method is of course next-generation sequencing (NGS). However, given the high cost, it is preferable to have some idea of what we have in hand before paying for NGS. Often this means identifying the insert sequence in the vector, and that identification is typically made by Sanger sequencing using primer sequences present in the vector backbone and flanking the insert. Therefore, a key step of vector analysis is identifying which vector backbone is present so the correct sequencing primers can be employed.

Most vectors in use today are derivatives and modifications of other, earlier, vectors. Sequences and features were added and removed to subserve various experimental purposes with time. This includes the addition or removal of sequences encoding selectable markers, promoters, terminators, tags, and sequences required for replication and expression in species other than E. coli. Thus, each vector backbone has its own unique “fingerprint” consisting of the set of sequence features incorporated over the course of its construction. From a practical point of view, this means that virtually all vectors in current use can be definitively identified by a panel of PCR reactions using primers designed against known sequence features. At Proxima, we use a panel of nearly 50 custom-designed primer pairs in a high-throughput qPCR-based assay to identify the sequence features of an unknown vector. Using this approach, most vector backbones are identified in a single step via the unique pattern of sequence features detected. Once the identity of the vector is known the insert sequence can be identified by the use of appropriate sequencing primers. Finally, one may invest in NGS for a complete and definitive vector sequence.
Professional help is only a few clicks away
So: don’t despair about the empty tube, the dead glycerol stock, or the smudged label! While this is necessarily a superficial treatment of the subject, we hope this post will set you along the path to confidently recovering those precious vectors relegated to the back of the fridge. If these steps feel like more than you want to undertake yourself, Proxima offers all these services at very reasonable rates that are typically lower in cost than for you to purchase all the required reagents and materials yourself.
Moreover, we have a “No DNA, No Pay” policy – so send us your empty tubes and dead glycerol stocks and if we can’t recover your vector then no there’s no charge. Either way, your vector mystery can be solved.
Use Proxima’s contact form to solve your mystery vector issues today.